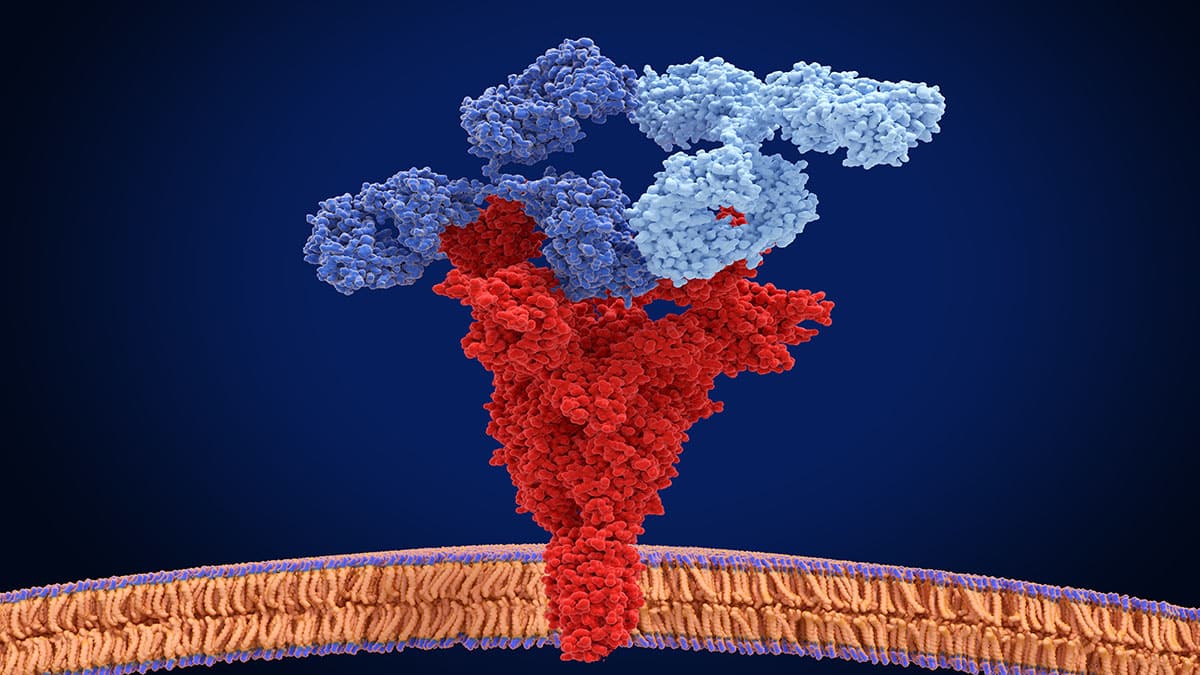
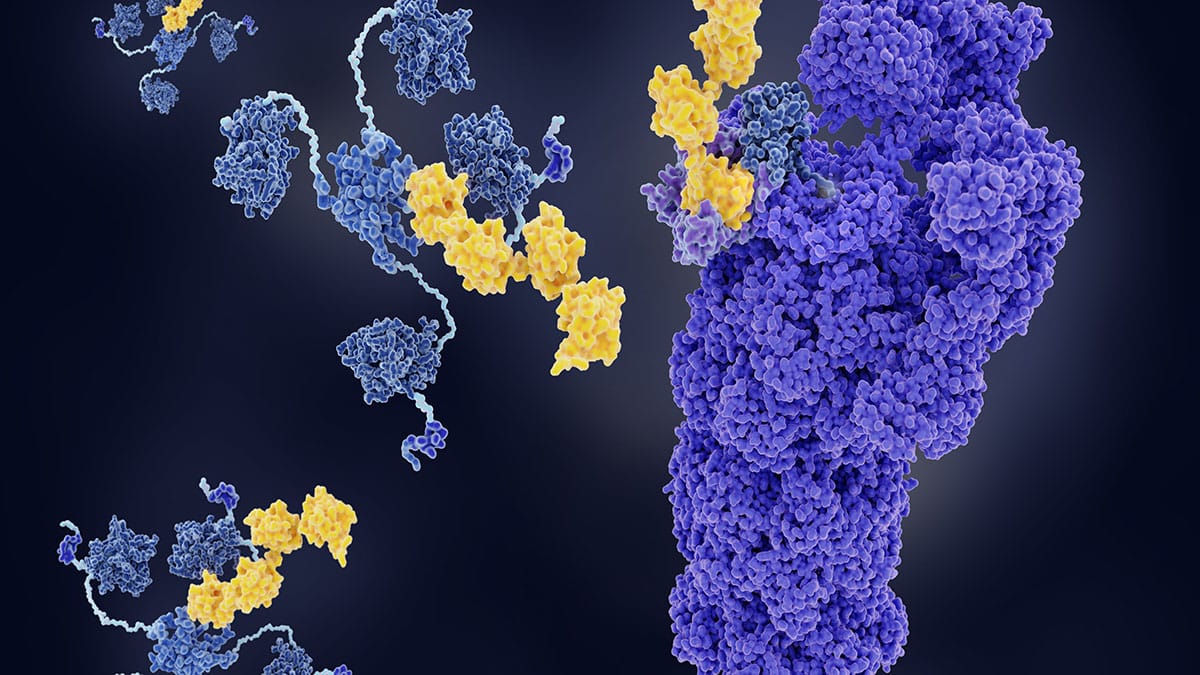
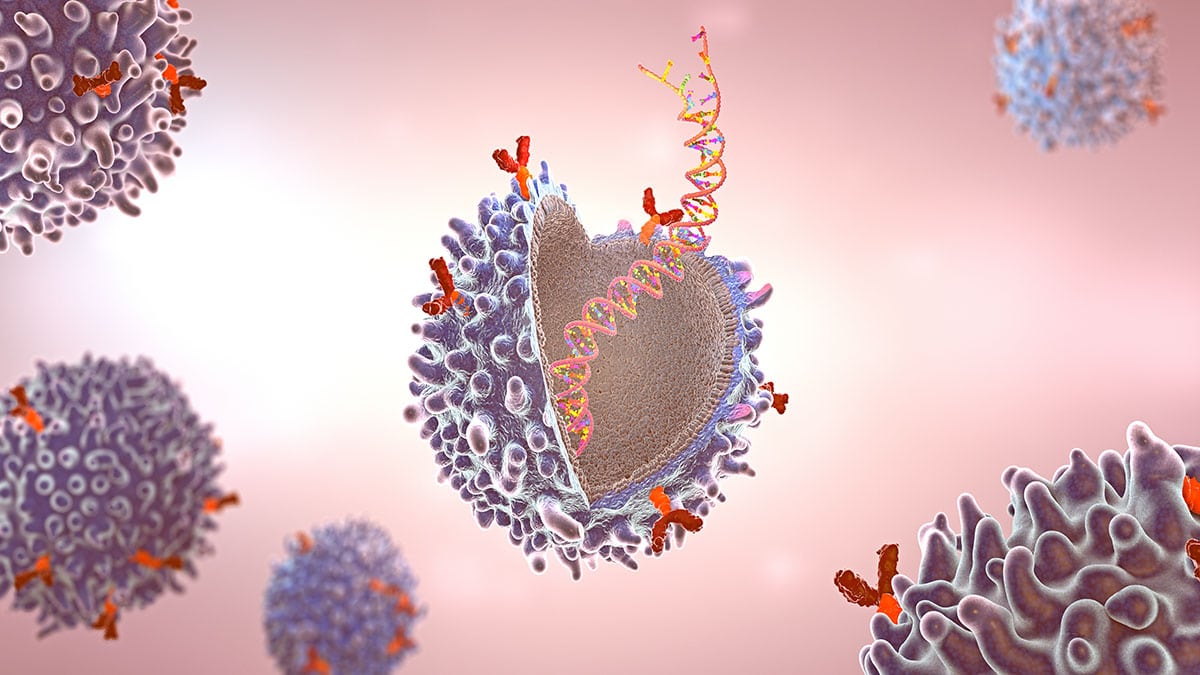
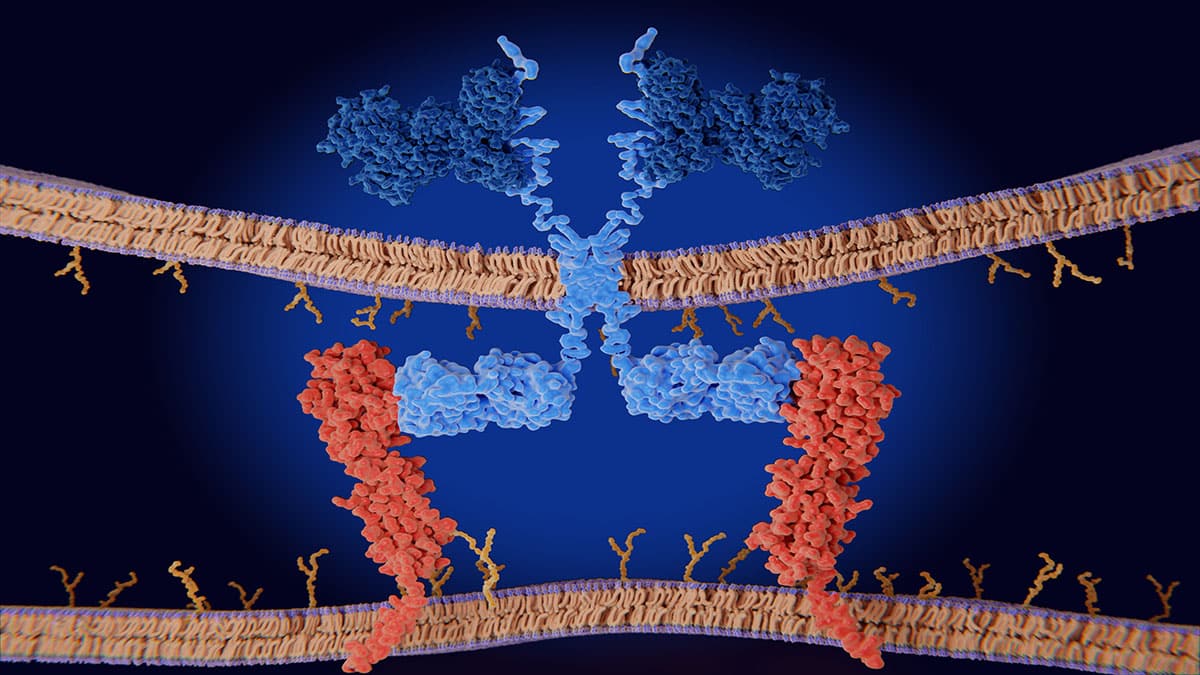
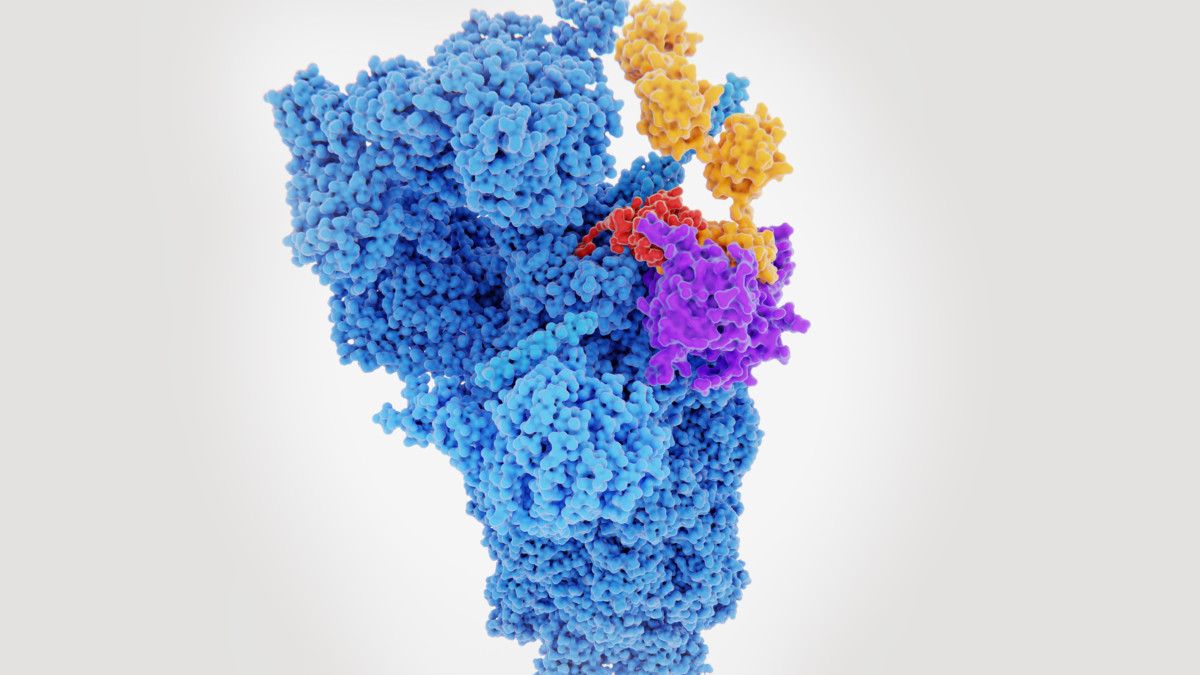
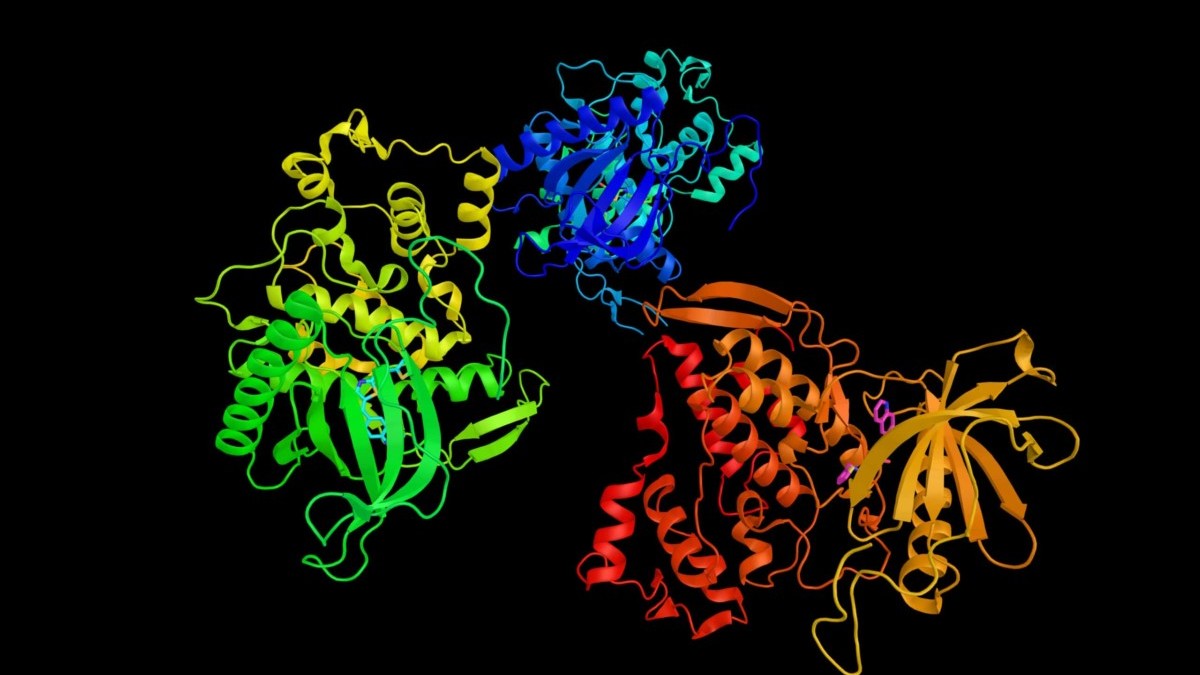
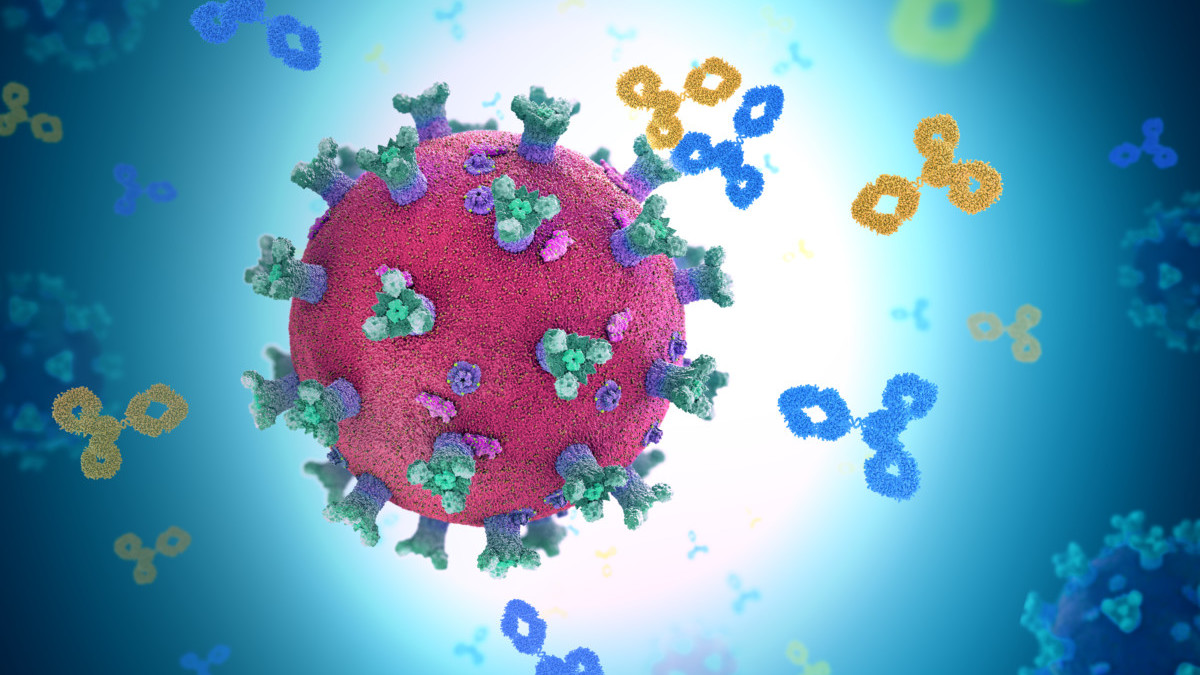
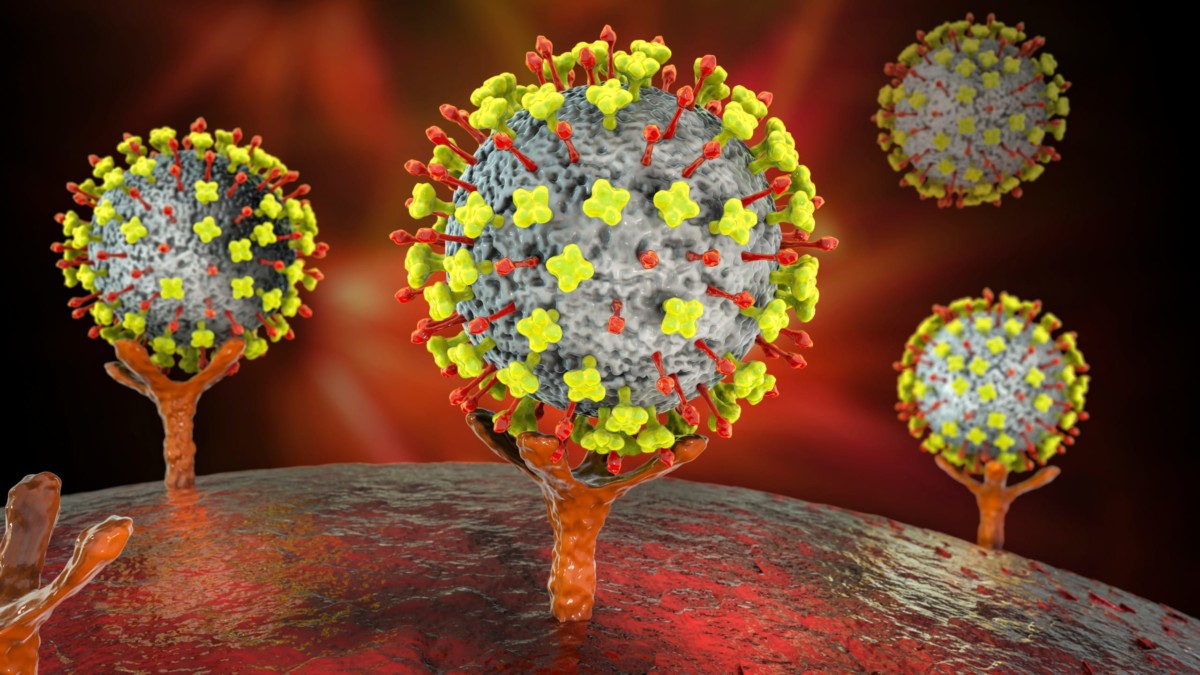
Major Histocompatibility Complex Therapeutic Applications
Written by: Jenna Kerry, MSc Published: February 26, 2025 Contents Introduction MHC Cancer Therapeutics MHC Autoimmune Disease Therapeutics MHC Infectious Disease Therapeutics MHC Therapeutics Computational Methods Immunopeptidomics Accelerating Immunopeptidomics with Rapid Novor's AI Algorithm Introduction The Major Histocompatibility Complex (MHC) is a key player in [...]