Glycan Analysis Service via LC-MS.
Discover the locations and relative quantities of common glycans using liquid chromatography and mass spectrometry.
How Glycan Analysis Works
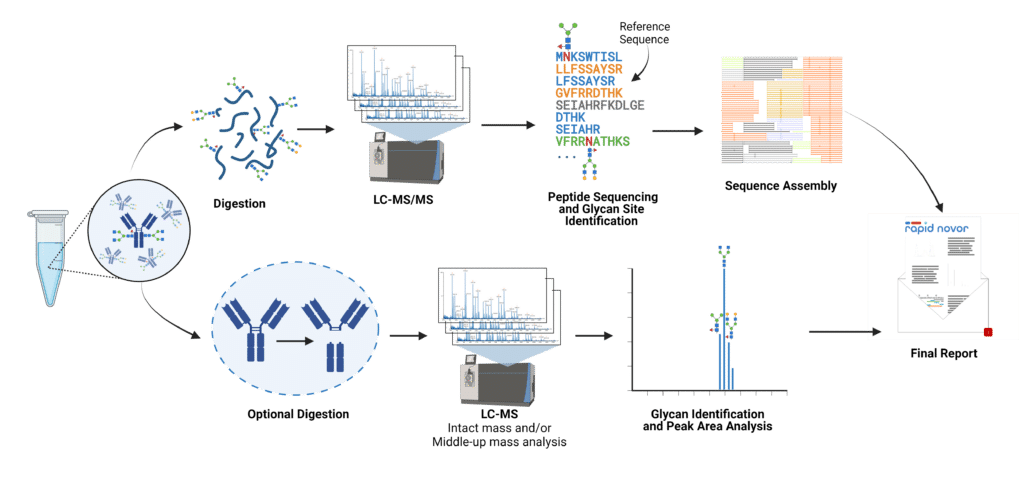
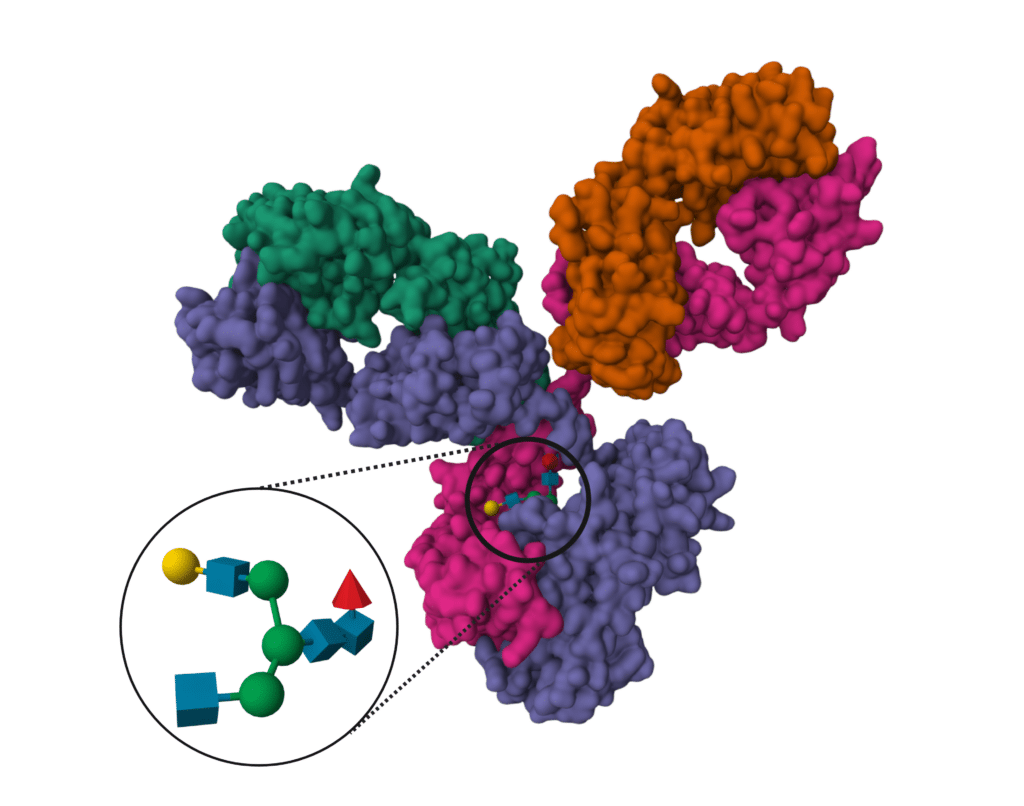
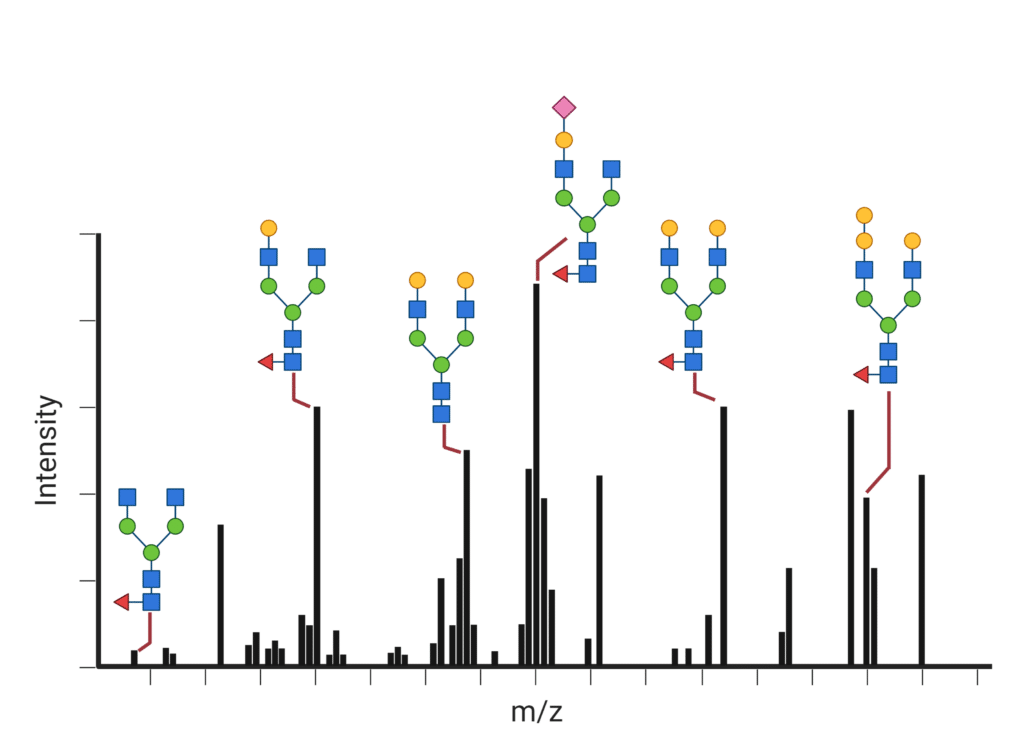
Glycosylation is one of the most common and functionally important post-translational modifications (PTM) that proteins experience. Each protein molecule with the same sequence of amino acids may be glycosylated in a different location or a different way. The most common glycoforms are well understood and are referred to by the number of terminal galactose residues and presence of fucose – (e.g. G2F is a Glycoform with 2 terminal galactose residues, and with a Fucosylated biantennary complex). The relative abundance of G0, G0F, G1, G1F, G2, G2F, etc. in a sample is referred to as the glycan profile. To determine the glycan profile and glycan sites, two types of experiment can be performed:
-
To determine the location of the glycosylation sites and to measure the glycan profile at a specific location, we digest the protein resulting in glycopeptides. Comparing the glycopeptides back to the amino acid sequence reveals the glycan sites, and comparing their peak areas reveals the glycan profile.
-
Measure the mass of the glycoproteins using liquid chromatography coupled with mass spectrometry to record the peak area for each glycoform. For antibodies, we can also digest with specific enzymes to analyze the regions (e.g. Fc and Fab) separately and derive the glycan profile at the subunit level.
Applications of Our Glycan Analysis Service
Glycan analysis can be used to understand a valuable research reagent, a wild type antibody, a recombinantly expressed antibody, or a marketed therapeutic. Glycan information, along with other biophysical characteristics can aid in selection and engineering of antibodies for therapeutics, IVD reagents, controls and research reagents. It can also be used to understand how glycosylation is applied by different expression systems, cell pools, cell lines and production conditions. This informs early decisions in production and is part of quality control and ICH Q6B standards.
The Rapid Advantage.
Identify Glycan Sites.
Fast and routine identification and confirmation of glycan sites informs your selection and protein engineering efforts. There are two types of glycans: n-linked glycans and o-linked glycans. N-glycan analysis is required for antibody development.
Streamlined Projects.
Reduce cycle time by adding glycan analysis to other services offered by Rapid Novor, including peptide mapping, de novo sequencing, transient expression, SPR and antibody discovery.
Talk to Our Scientists.
We Have Sequenced 10,000+ Antibodies and We Are Eager to Help You.
Through next generation protein sequencing, Rapid Novor enables reliable discovery and development of novel reagents, diagnostics, and therapeutics. Thanks to our Next Generation Protein Sequencing and antibody discovery services, researchers have furthered thousands of projects, patented antibody therapeutics, and developed the first recombinant polyclonal antibody diagnostics.
Talk to Our Scientists.
We Have Sequenced 9000+ Antibodies and We Are Eager to Help You.
Through next generation protein sequencing, Rapid Novor enables timely and reliable discovery and development of novel reagents, diagnostics, and therapeutics. Thanks to our Next Generation Protein Sequencing and antibody discovery services, researchers have furthered thousands of projects, patented antibody therapeutics, and ran the first recombinant polyclonal antibody diagnostics
Talk to our scientists. We have sequenced over 9000+ antibodies and we are eager to help you.
Other Antibody Characterization Services.
Peptide Mapping
Match the masses of individual peptides to the expected masses from a given sequence, maximizing sequence coverage and confidence.
Explore our LC-MS Peptide Mapping Service
Intact Mass Analysis
Determine the mass of the whole antibody or its sub-units to explore a variety of characterization applications.
Explore our Antibody Intact Mass Analysis Service
Glycosylation Analysis
Find glycosylation site occupancy, and profile their relative abundance %. Compare many samples.
Explore our Glycosylation Analysis LC-MS Service
Rapid PTM Analysis
Locate, identify and quantify post-translational modifications. Compare between samples.
Explore our PTM Analysis LC-MS Service
Sequence Variant Assessment
Identify and quantify variants of your intended antibody within your sample, and consider other contaminants.
Explore our SVA Analysis LC-MS Service
Disulfide Bond Analyses
Analyse peptides cross linked by disulfide bonds. Quantify disulfide bond shuffling in engineered constructs.
Explore our Disulfide Bond Analysis LC-MS Service
DAR Analysis
Determine the ratio of antibody to payload in ADC development.
Explore our Drug-Antibody Ratio Analysis Service
Antibody Sequencing, Discovery and Characterization Services.
Antibody Sequencing & Discovery Services.
REmAb® mAb Sequencing
Monoclonal antibody sequencing from small antibody samples, no need for hybridoma or DNA information. Full sequence in record time.
Explore Antibody Sequencing Services
REpAb® pAb Sequencing
Polyclonal antibody sequencing and antibody discovery service. Sequence antibodies from blood or a polyclonal mixture.
Explore Antibody Discovery Services
RapidSPR™ Analysis
Label-free evaluation of antibody-antigen binding using surface plasmon resonance on Biacore or Nicoya. Can be employed for detailed kinetic profiling, kinetics screening, or epitope binning.
Explore SPR Services
RapidHDX-MS™
Mass spec based epitope mapping. Epitope mapping for identify the binding site of an antibody to its corresponding antigen with the highest confidence and resolution.
Explore HDX-MS Epitope Mapping Service
MATCHmAb™
Antibody verification by rapid peptide mapping. Antibody sequence confirmation and full detailed mapping for development purposes.
Explore Peptide Mapping Service
Antibody Characterization
LC-MS analysis for study of antibody glycosylation, disulfide bonds, post-translational modifications, sequence variants, intact mass and size.
Explore Antibody Characterization Services





