Written by Yuning Wang, PhD
Written by Yuning Wang, PhD
September 14, 2021
What are Amino Acids?
Amino acids are small organic molecules that make up peptides and proteins. All living organisms share the same set of amino acids. Amino acids come together in different orders (sequences) to form proteins. As such, each type of protein has a different three-dimensional structure and biological activity.
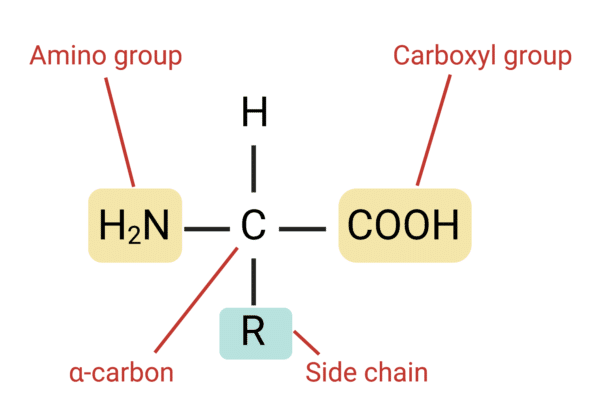
Structure of Amino Acids
All amino acids possess a carboxylic acid group (-COOH) and an amino group (-NH2), both linked to their α-carbon atom. A side chain (R group) is also attached to the α-carbon. Side chain structures vary, each donning specific biophysical properties to their amino acid.
Like sugar, all amino acids (except glycine) are stereoisomers that exist in D and L-forms. But almost all naturally occurring amino acids found in proteins are L-forms (with a few exceptions, such as D-amino acids found in bacterial cell walls, antibiotics, and hormones). Why L-form amino acids are favorable in nature still remains an evolutionary mystery.
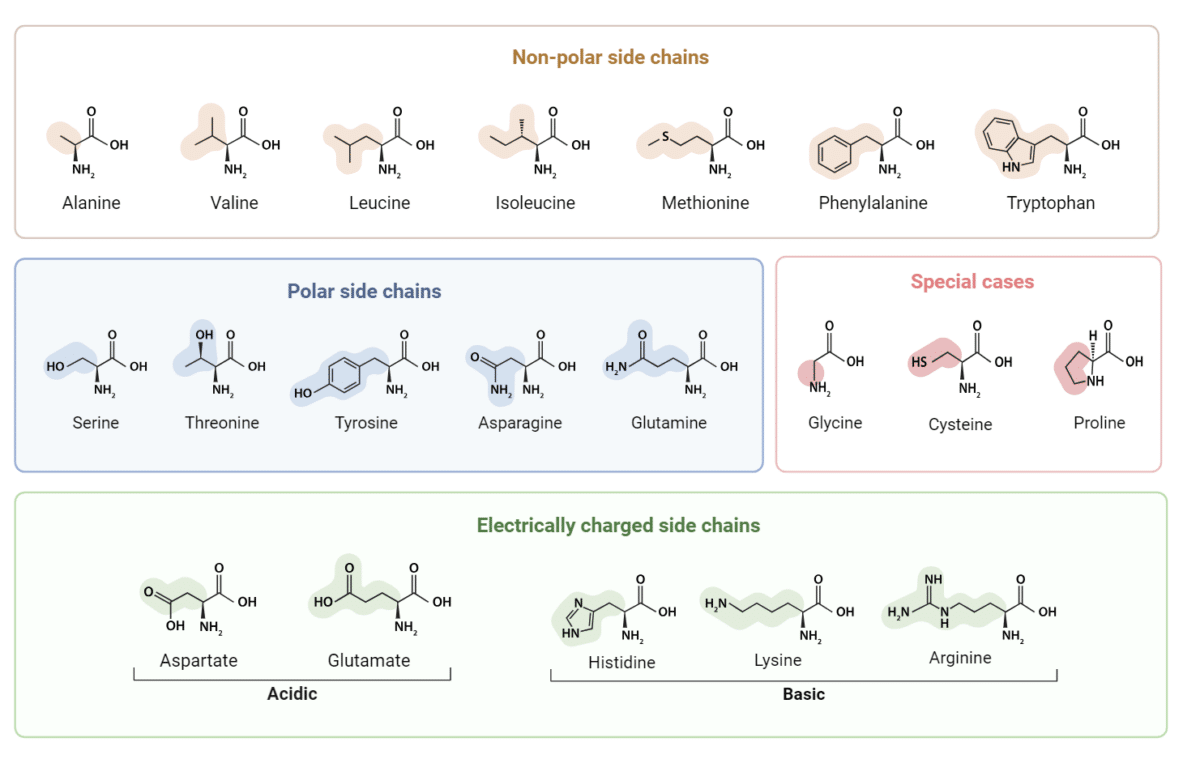
Types of Amino Acids
DNA encodes twenty types of amino acids that make up proteins in all organisms, from bacteria to plants, to animals. These amino acids can be broadly grouped based on their biophysical properties (e.g., charge, hydrophobicity, and polarity of their side chain). The properties of the side chains govern the way they interact with surrounding molecules, and consequently impact the protein’s 3D structure.
Non-polar Amino Acids
Non-polar amino acids carry a hydrophobic side chain, and are commonly located in the interior of proteins, whereas polar amino acids are typically found on protein surfaces due to the hydrophilic properties of their side chains.
Polar and Electrically-charged Amino Acids
Among the 20 amino acids, five have a side chain that can be electrically charged at physiological pH. Two are negatively charged (acidic) and three are positive (basic). These charged amino acids play important roles in stabilizing protein 3D structures, and mediating protein-ligand interactions via electrostatic attractions.
Special Amino Acids
Nonetheless, there are three amino acids that stand out for their unique properties: (1) Glycine is the simplest amino acid and it doesn’t really have a side chain. It has a hydrogen as its R group giving glycine much flexibility to fit in different parts of proteins. (2) Cysteine has a reactive sulfur-containing thiol group that can form disulfide bonds between two cysteine residues. (3) Proline incorporates the backbone into its side chain to form a cyclic ring, making it unable to adopt many of the main chain conformations. It can also introduce kinks to occur in a protein chain.
Non-standard Amino Acids
Besides the 20 standard amino acids, many non-standard amino acids are also found in cells, which are typically chemically modified after they are incorporated into a protein. These chemical modifications are part of post-translational modifications. Examples of these non-standard amino acids include carboxyglutamic acid (a product of glutamine carboxylation), hydroxyproline (a product of proline hydroxylation), methyllysine (a derivative of lysine where its side chain has been methylated) and more. These modifications result in important protein properties that enable and improve a wide range of biological functions.
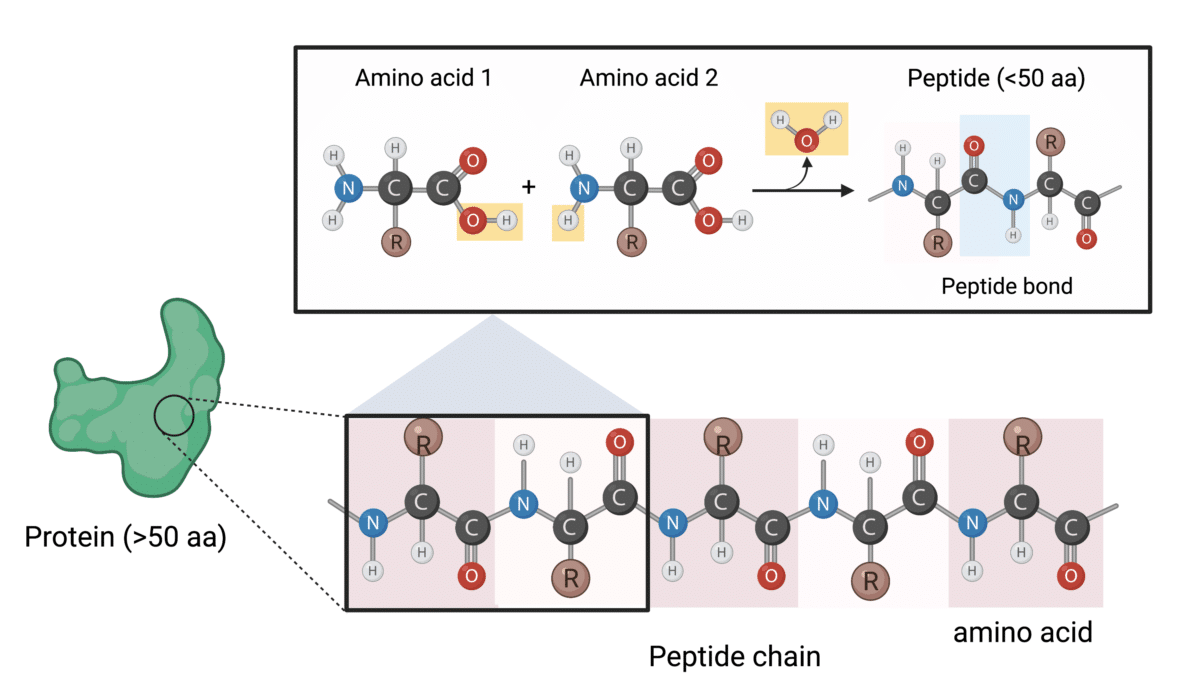
Peptide Bonds
Amino acids are commonly joined together by an amide linkage, called a peptide bond. The peptide bond is formed by a condensation reaction between two amino acids where one loses a hydroxyl from its carboxyl group (-COOH), and the other loses a hydrogen from its amino group (-NH2). Hence, this reaction is accompanied by the loss of a water molecule. Therefore, the backbone of a chain of amino acids (or a peptide) contains repeated core atoms (–N–C–C–), and a variety of side chains projecting from the backbone.
Peptide, Polypeptide Chains, and Proteins
Peptides are short polymers joined by two or more amino acids, usually fewer than 50 amino acids long. In cells, peptides can perform various biological functions. For instance, neuropeptides act as transmitters in the nervous system, and peptide hormones like insulin contribute to the maintenance of homeostasis in living organisms.
A polypeptide is generally considered a linear chain of many amino acids. A polypeptide that contains more than approximately 50 amino acids is known as a protein. Proteins can be formed from one or more polypeptides joined together.
How is the Sequence of Amino Acids Determined?
It is estimated that the human body can produce between 10,000 to several billion different proteins. Each type of protein differs in its number and sequence of amino acids, and knowledge of the amino acid sequence is essential for the understanding of its three-dimensional structure, cellular location, function, and evolution.
The sequence of amino acids can be determined using different methods, such as Edman degradation and mass spectrometry-based de novo amino acid sequencing. Check out our blogs to learn more about these methods:
Protein Sequencing Service
Rapid Novor offers de novo protein sequencing service and polyclonal antibody sequencing using liquid-chromatography with mass spectrometry to identify the amino acid sequence of proteins without access to genomic information.
Talk to Our Scientists.
We Have Sequenced 10,000+ Antibodies and We Are Eager to Help You.
Through next generation protein sequencing, Rapid Novor enables reliable discovery and development of novel reagents, diagnostics, and therapeutics. Thanks to our Next Generation Protein Sequencing and antibody discovery services, researchers have furthered thousands of projects, patented antibody therapeutics, and developed the first recombinant polyclonal antibody diagnostics.
Talk to Our Scientists.
We Have Sequenced 9000+ Antibodies and We Are Eager to Help You.
Through next generation protein sequencing, Rapid Novor enables timely and reliable discovery and development of novel reagents, diagnostics, and therapeutics. Thanks to our Next Generation Protein Sequencing and antibody discovery services, researchers have furthered thousands of projects, patented antibody therapeutics, and ran the first recombinant polyclonal antibody diagnostics