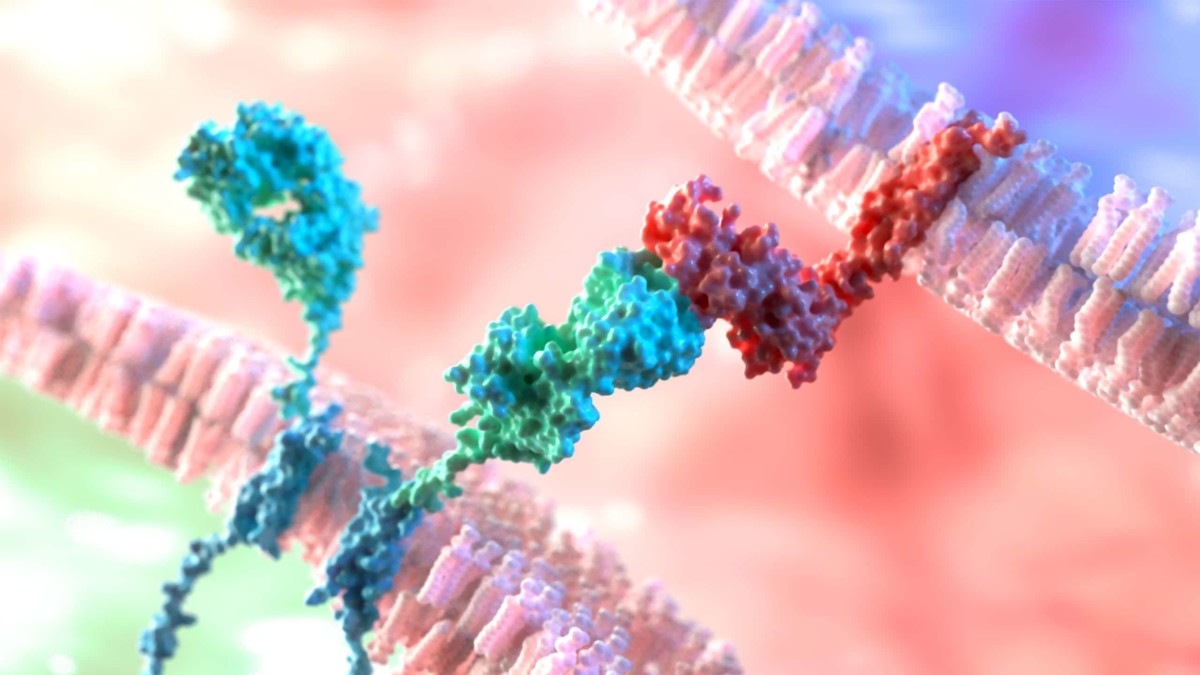
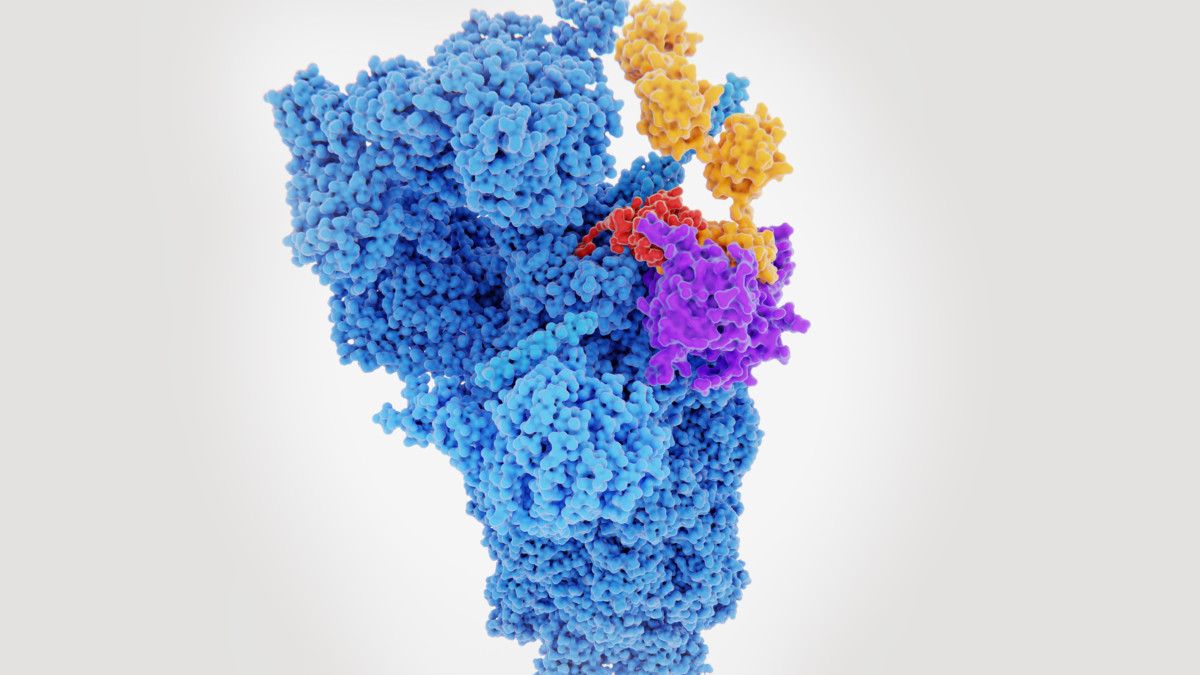
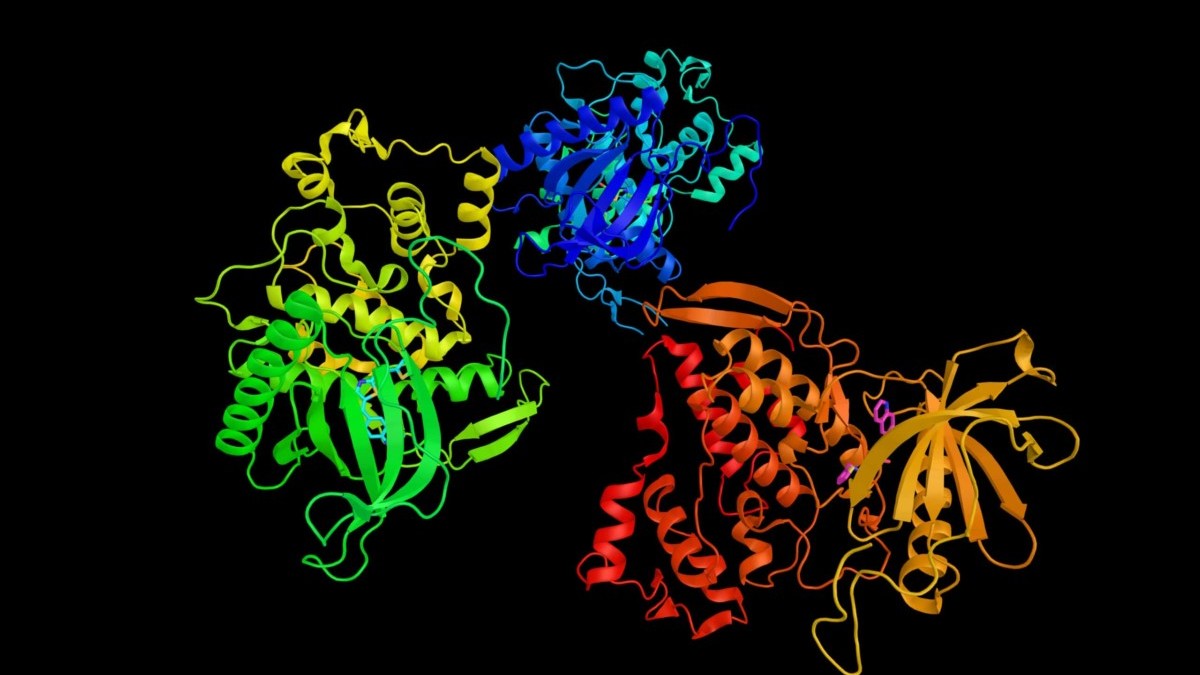
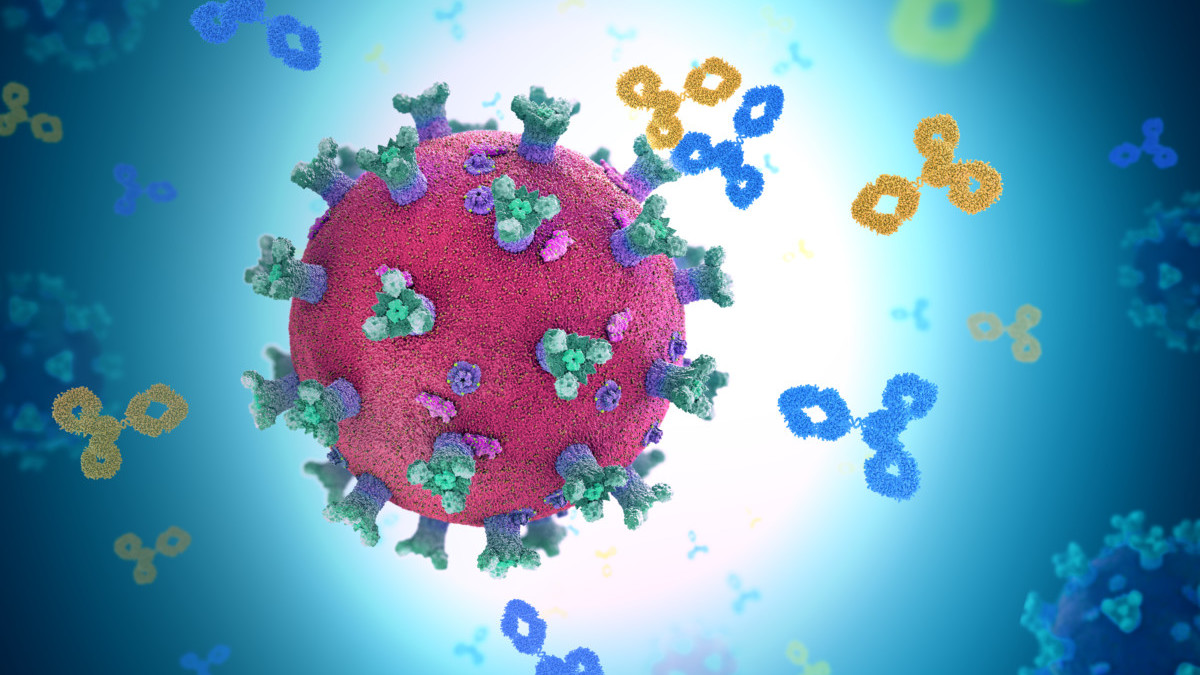
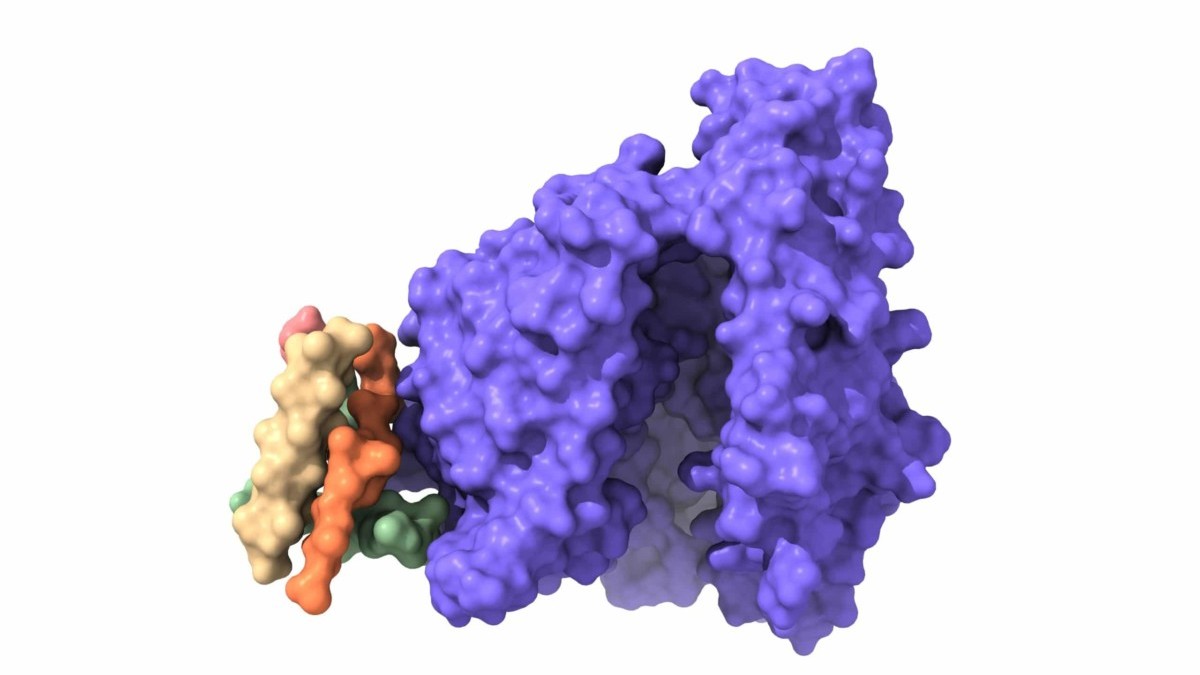
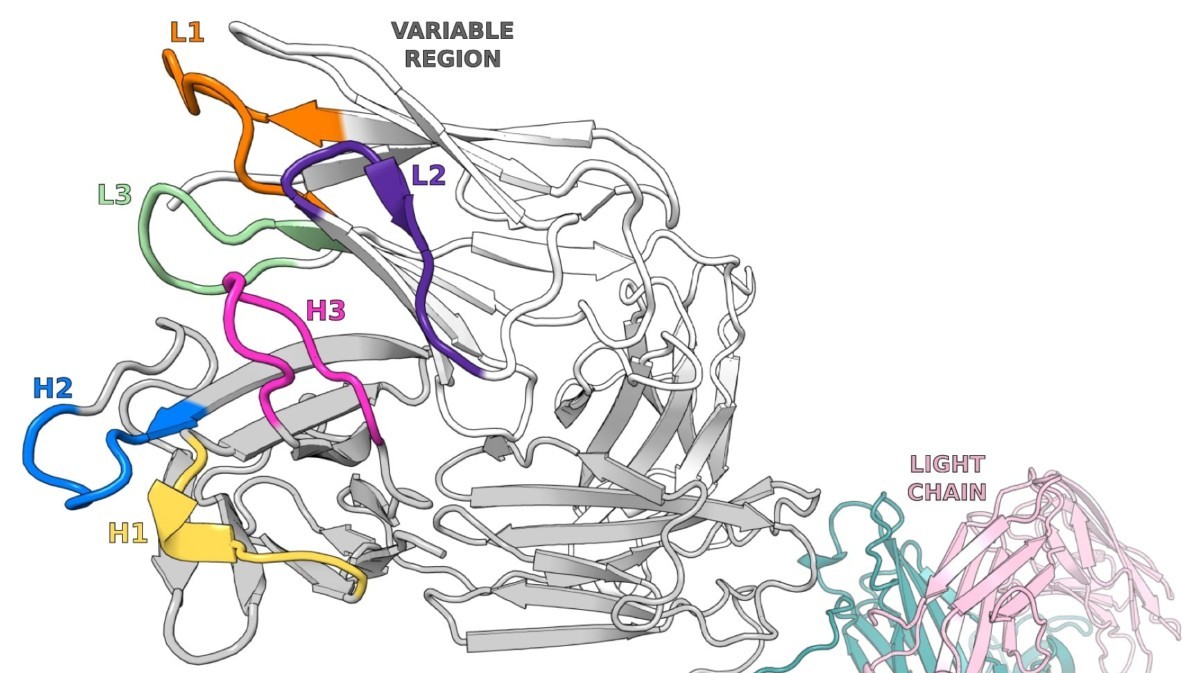
Antibody Discovery Processes
Written by: Vanessa Yoon Calvelo, PhD Published: June 6, 2023 Updated: July 7, 2024 Contents Introduction Early Discovery and Development of Therapeutic Antibodies Target Identification and Validation Antibody Discovery and Expression Lead Characterization and Selection Lead Engineering and Optimization Candidate Selection and Downstream Characterization Introduction [...]