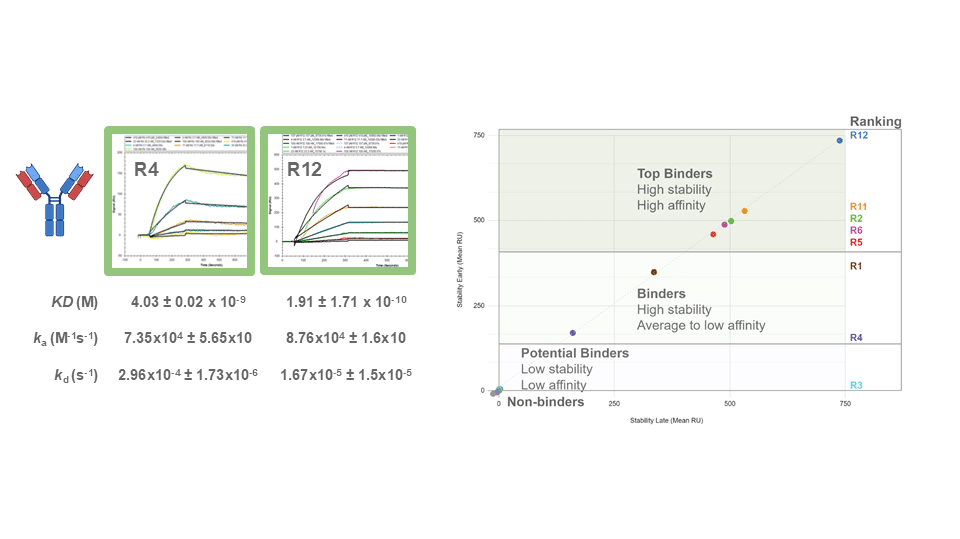
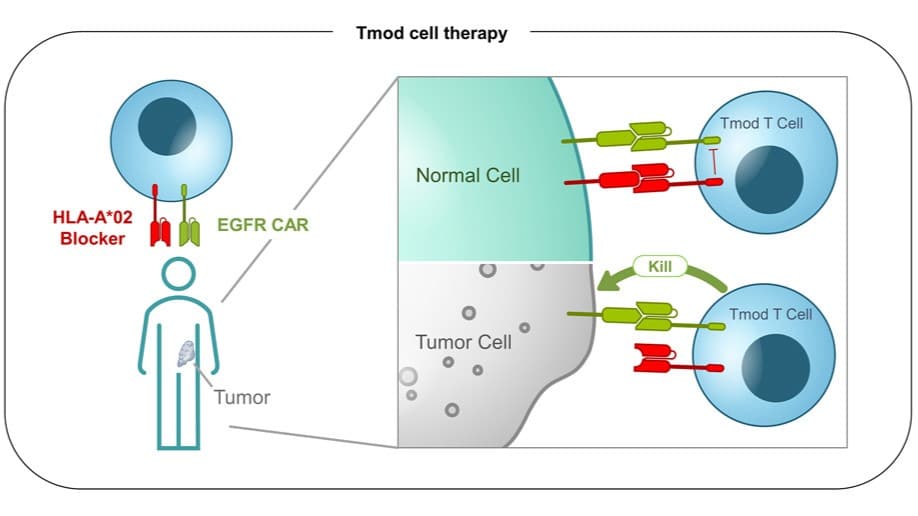
Biparatopic Antibodies
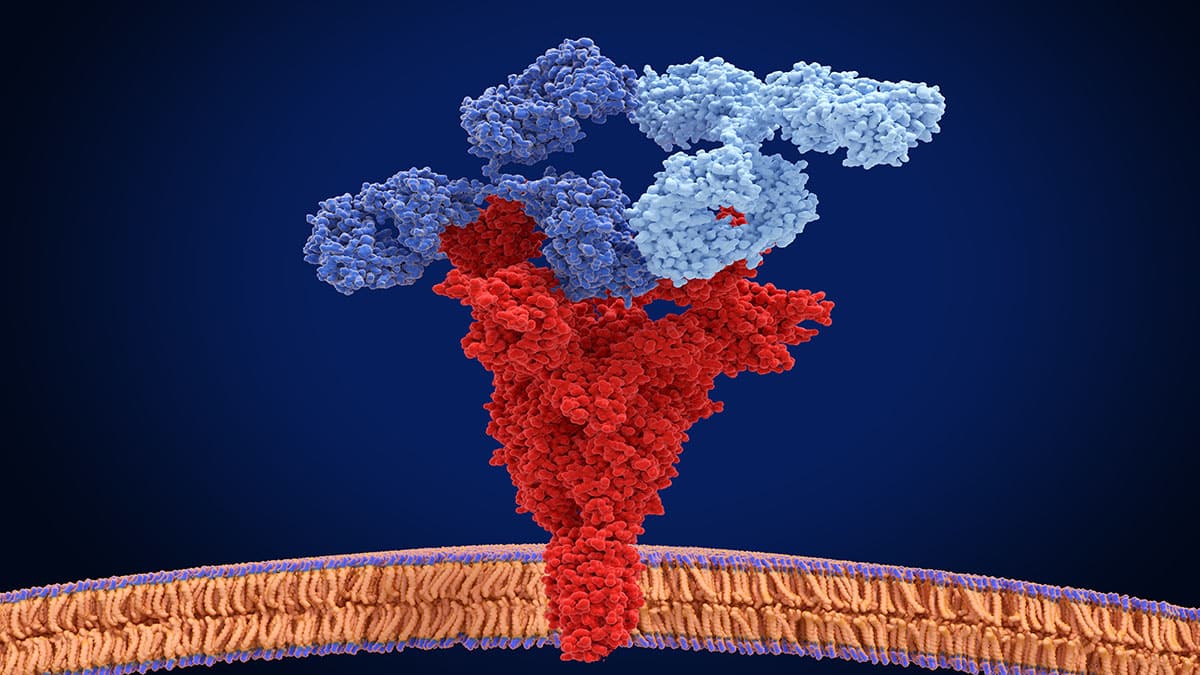
Written by: Genya Gorshtein, MSc Published: June 13, 2025 Contents Introduction What Are Biparatopic Antibodies? How Are Biparatopic Antibodies Engineered? Biparatopic vs. Bispecific Antibodies: What’s the Difference? Engineering Biparatopic Antibodies with Rapid Novor Introduction Biparatopic antibodies are a subclass of bispecific antibodies that bind [...]